

 |
I. Ya. Grichanov All-Russian
Institute of Plant Protection, Podbelskogo 3, St. Petersburg, Pushkin
189620, Russia: e-mail: grichanov@mail.ru
|
Dolichopodidae (Diptera: Empidoidea)
I. Ya. Grichanov
All-Russian Institute of Plant Protection, Podbelskogo 3, St. Petersburg, Pushkin 189620, Russia: e-mail: grichanov@mail.ru
BIOGEOGRAPHY
The generic and specific names of predatory flies of the epifamily Dolichopodoidae (Diptera) belong to 257 extant and 30 fossil genera of Dolichopodidae (including 7 extant and 2 fossil genera of Parathalassiinae), 2 extant and 6 fossil genera of Microphoridae, 8063 valid species names (including about 120 fossil species) belonging to the families Microphoridae and Dolichopodidae including the subfamily Parathalassiinae. The total number of Dolichopodoidae increased by nearly 300 species since 2014 till 2017.
At present a total of about 800 Afrotropical species are known to occur. Many species described earlier this century were incompletely described, some lacking male descriptions or were inadequately illustrated. Authors, in their descriptions, often omitted important characters regarded today, as having generic level value. For this reason no less than 20 species are waiting to be synonymized or to be declared Nomina Dubia in future revisions of type material. Several hundred species probably await description. The majority of the Afrotropical species were described by Th. Becker, C.H. Curran, I.Ya. Grichanov, C.G. Lamb, O. Parent and P. Vanschuytbroeck (see Dyte & Smith 1980; Grichanov 1997-2017).
The fauna of the Congo basin (Kinshasa) may be regarded well-known, while fragmentary investigations have been undertaken of the fauna of South Africa, Madagascar, Tanzania, Uganda, Nigeria, Kenya and Cameroon. Adjacent African islands and other Afrotropical countries have been poorly studied.
BIOLOGY AND ECONOMIC SIGNIFICANCE
Larvae occur in mud, damp soil, leaf litter, moss, algal mats, decaying marine algae, sap wounds, under bark, in tree hole debris, within plant tissues, and on intertidal rocks (Dyte, 1959; Poulding, 1998, 2011). Most larvae are thought to be predators, or scavengers, except for Thrypticus which are phytophagous stem-miners in grasses, sedges and rushes (Dyte, 1959; Poulding, 1998).
Most of the numerous species of the cosmopolitan genus Medetera are associated with tree trunks. Imagoes of some Medetera species may be encountered in montane regions on large stones and rocks covered by mosses and lichens and in semi-desert regions in and around rodent burrows and other ground cavities. Many scientific works describe value of long-legged flies in forestry, first of all, value of flies of the genus Medetera as highly effective predators of cryptic stem and bark pests. In Russia in 1960-s, the first attempts to their breeding and use for control of bark beetles and other harmful coleopterans were made. Application of long-legged flies and other entomophages in forestry practice as bioagents has not found way to practice, probably, by economic reasons. Regulatory role of Medetera species has been studied in Estonia, Georgia, Latvia, Lithuania, Russia, Ukraine, USA and many other countries. Meanwhile, dolichopodids have a great importance for agriculture. They have been recorded in considerable amounts in orchards (apple, pear, peach), vineyards, winter and spring wheat, evidently being a stable component of these agroecosystems (Grichanov, 1990, 1991, 1997; Negrobov & Kamolov, 1992; Grichanov & Shamshev, 1993). Medetera flies can also feed on aphids, thrips and mites populating plants of cotton, grain, vegetable, fruit and other cultures, together with other entomophages regulating development and reproduction of dangerous pests, especially at irrigation farming (Rathman et al., 1988; Meuffels et al., 1989; Brunel et al., 1989; Grichanov & Shamshev, 1993).
Artificial breeding of predatory long-legged flies and their application in the closed ground for control of hothouse pests seem to be rather promising. First such experiments with the Medetera flies have been undertaken by the Italian researchers (Moreschi, 2001, 2002a,b; etc.). They have underlined, that Medetera feed in hothouses and greenhouses on such harmful invertebrates, as sciarid midge Bradysia paupera, aphids Macrosiphum rosae, Aphis fabae, Myzus persicae, Illinoia liriodendri, whiteflies Trialeurodes vaporariorum, Bemisia tabaci, thrips Frankliniella occidentalis, springtail Folsomia candida and mites (Tyrophagus spp.). Three methods of Medetera cultivation, from rather complex and laborious to the most simple, have been tested. Having carried out the detailed analysis of biological characteristics of bred and released in hothouses Medetera flies, the Italian experts have noted both prospects and economic efficiency of the proposed method of biological control. We recommend all other experts in the field of biomethod application in the closed ground to pay attention to this very interesting group of insect entomophages.
Several genera have
been reported as predators on the immatures and emerging adults of mosquitoes,
including the Anopheles gambiae Giles complex, the most important vectors
of malaria in sub-Saharan Africa (Service, 1971, 1973; Ulrich, 2005). Several
Neotropical species of the phytophagous genus Thrypticus have been reared
from the petioles and stems of water hyacinth. These species have potential
as biocontrol agents of this New World aquatic plant which is an invasive
weed in the waterways of Asia, Australasia and Africa (Bickel & Hernández,
2004). A number of studies (e.g., Gelbič & Olejníček, 2011 and references
cited therein) demonstrate that dolichopodids have very specific habitat
requirements and react quickly to environmental changes, making them potentially
useful as bioindicators for site quality assessment and conservation purposes.
In addition to feeding
on live prey, many adult dolichopodids have also been observed taking up
honeydew (Ulrich, 2005) and some are known to be anthophilous and feed
on nectar (Pollet et al., 2010 and references cited therein).
Most adult dolichopodids occur on sand, damp ground, grass, leaves, tree trunks, river rocks, and on other surfaces near open water. Adults are abundant in warm moist environments, including stream and lake margins, forests, salt marshes, seashores and freshwater seepages. A great many species of Dolichopodidae may be collected by use of Malaise and light traps, or by sweeping through vegetation with a net.
Many rare species are known only from their type localities. When such species are only known from small blocks of remnant or disturbed vegetation, their long-term survival is more problematical, especially in highly altered agricultural and urban districts. They may be threatened, if their remnant habitats are degraded by burning, grazing, clearing or invasion by exotic weeds, replaced by settlements, roads and other anthropogenic landscapes. Active application of chemical plant protection means, land reclamation, changes in land use technologies towards more rationalized approaches can have adverse effects on the biodiversity of natural ecosystems.
[Originally] by D.J. Bickel & C.E. Dyte
Flies of the family Dolichopodidae are often abundant in moist habitats and are frequently found on foliage, tree trunks, mud flats, and river rocks. Adults are predaceous on soft-bodied invertebrates and serve as important general control agents of many pest species.
The maggotlike larvae are found in such habitats as soil, rotted vegetation, mud, and under bark. Most are predators or scavengers, although the larva of Thrypticus is a phytophagous stem miner in various Monocotyledons [see Dyte (1967) for review of immature stages]. Williams' (1938b,c, 1939) studies of the immature stages of Hawaiian dolichopodids are among the best documented for the family.
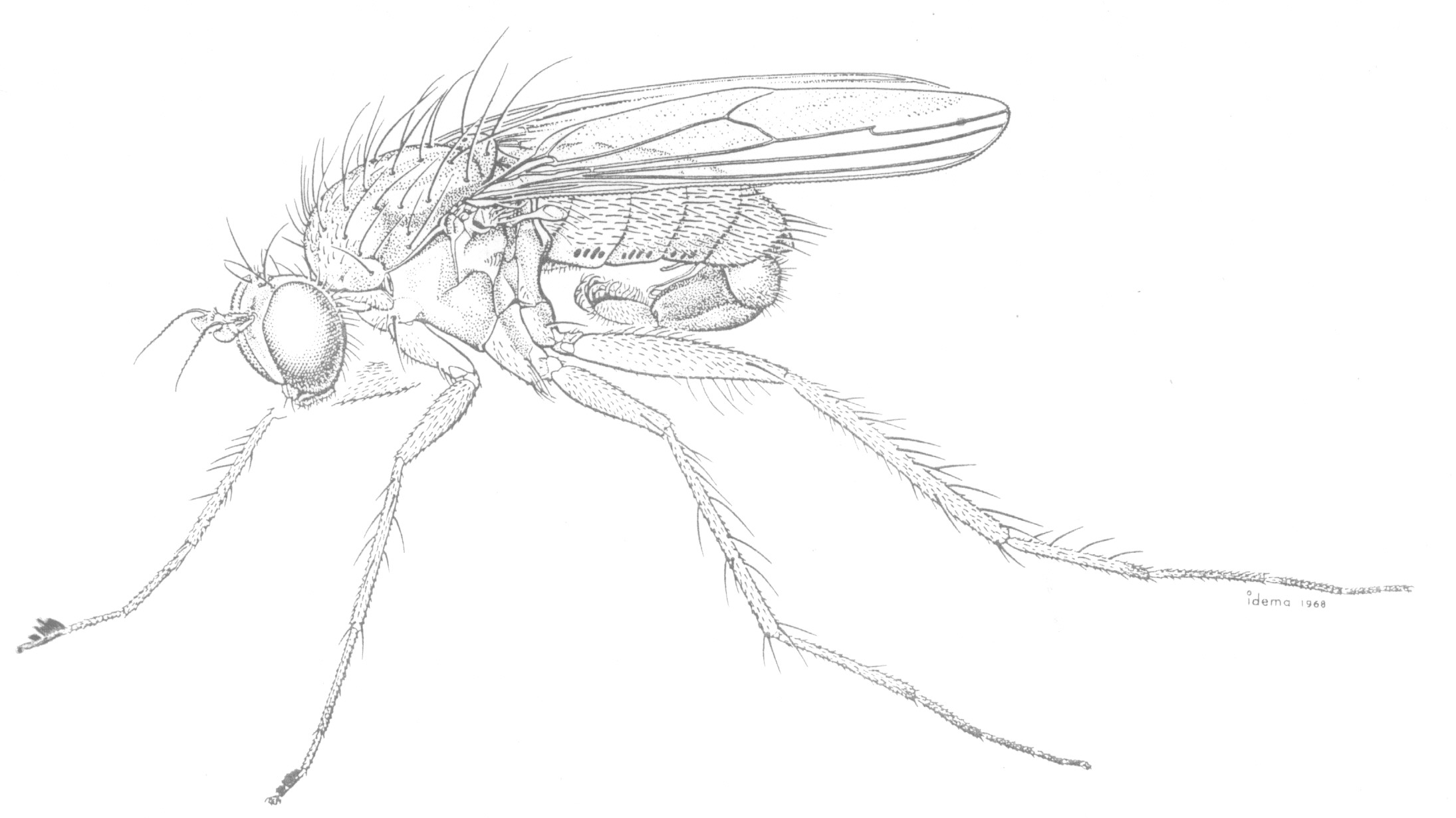
The elaborate secondary sexual characters of male dolichopodids are assumed to aid in species recognition during courtship and often show parallel development in unrelated groups. These characters include flaglike flattening of the arista and tarsi, strongly modified setae and cuticular projections, prolongation and deformation of podomeres, orientated silvery pruinosity, maculation and deformation of wings, etc. The hypopygium is often greatly enlarged and exposed and usually carries species-specific characters. The tiny Babindella, of the endemic Australian subfamily Babindellinae, shows secondary postabdominal symmetry (Bickel 1987b).
The Dolichopodidae is one of the most diverse families of flies, with approximately 5,000 described species. In the Australasian/Oceanian Regions, 995 species representing 77 genera in 9 subfamilies are currently known, although many new species and genera await description, especially from Australia and the Melanesian Archipelago. Apart from southern temperate elements in New Zealand and Australia, much of the Australasian/Oceanian fauna has strong affinities with that of the Oriental Region. A notable exception is the presence of the Neotropical Condylostylus longicornis in eastern Polynesia.
Some "tramp" species, such as Medetera grisescens and Chrysosoma leucopogon, show extraordinarily widespread distributions throughout both the Oriental and Australasian regions, including many isolated oceanic islands. Several brachypterous species are known from subantarctic islands. Of particular interest is the extensive speciation of Campsicnemus in the Hawaiian Islands (138 spp., with many more awaiting description). This radiation invites comparison with that of the well-studied Hawaiian Drosophila.
The subfamily arrangements followed here are based on Ulrich (1981), with modifications. The limits of certain subfamilies, such as the Diaphorinae and Sympycninae are unclear and some genera, such as Sympycnus, are polyphyletic assemblages. Recent revisionary studies on the Medeterinae (Bickel 1986a,b,c, 1987c) and Hydrophorinae (Grootaert & Meuffels 1984, Meuffels & Grootaert 1984) have added to the regional understanding of these taxa.
Becker's (1922b) monograph, although dated, remains the basic reference for both the Oriental and Australasian/Oceanian regions. Important faunistic treatments include those on the Hawaiian Islands (Hardy & Kohn 1964, Tenorio 1969), New Zealand (Parent 1933b), Australia (Hardy 1930a, 1935b, 1939c; Parent 1932a, 1933a), Samoan Islands (Lamb 1929), and New Guinea (de Meijere 1906b, 1913a,b, 1915b; Parent 1939a). Additional species descriptions are found in the many papers of Parent. Robinson & Vockeroth (1981) provided a well-illustrated general review of the family.
Family DOLICHOPODIDAE
Dolichopodids, commonly called long-legged
flies, are found worldwide and consist of over 5,000 species in some 150
genera. Adults and most larvae are predaceous on other insects and small
invertebrates. Adults of extant forms inhabit moist, cool, sylvan habitats
near water sources where they can be found resting on tree trunks, vegetation,
or skating on the water surface in pursuit of prey items. Larvae are found
in rotting vegetation, wet soil, and under bark where they are scavengers
or predators.
The Dolichopodidae are well represented
in the fossil record, especially as amber inclusions. Although only Baltic
amber material of this family has been described to any extent, the family
is present in most other faunas as well (Spahr, 1985). Many adults of this
family are tree associates and patrol up and down trunks in search of both
mates and food. Hence, they are especially susceptible to being entrapped
in tree resins and fossilized in amber. Modern trunk-associated dolichopodids
include: most Medeterinae (especially Medetera Fischer von Waldheim
and Systenus Loew), all Neurigoninae, Achalcus Loew, many
Sciapodinae (especially Sciapus Zeller s. str.), and some Diaphorinae.
All of these taxa also occur in Baltic amber.
As seen in Baltic amber faunas generally
(Larsson, 1978), most recent insect families had radiated by the Early
Tertiary. This is the case with the Dolichopodidae, and genera appear well
defined and essentially modern at that time. However, the Baltic amber
fauna described by Meunier is badly in need of revision, especially in
light of large unworked collections of fossils in many institutions. Particular
attention must be paid to Meunier's generic concepts and species placements.
In addition to the determined material listed below, undetermined material of this family has been recorded from the Upper Cretaceous ambers of Canada (McAlpine & Martin, 1969: 837) and Siberia (Zherichin & Sukacheva, 1973: 18, 37), the Oligocene/Miocene amber of the Dominican Republic, and Miocene amber of Chiapas, Mexico (Poinar, 1992), the Lower Miocene deposits of Sado Island, Japan (Fujiyama, 1985: 43), the Miocene Bitterfeld amber of Germany (Schumann & Wendt, 1989), and the Eocene/Oligocene deposits of the Bembridge Marls on the Isle of Wight (Jarzembowski, 1976: 12).
Ref.: Meunier (1907cj,1908ad, f, review of Baltic amber taxa).